SOMERSET, N.J., April 14, 2025 – CIMS Global proudly announces the release of DMC-HUB 1.0, a comprehensive, secure, and centralized digital environment designed to support the evolving needs of Data Monitoring Committees (DMCs). In response to latest FDA guidance on the Use of DMC’s in Clinical Trials, as well as and industry trends toward adaptive trial designs, multiregional oversight, and program-level monitoring, DMC-HUB offers a fully integrated suite of tools to enhance the safety, efficiency, and scientific integrity of clinical trial oversight.
“We built DMC-HUB to address the increasing complexities of clinical trials, helping sponsors by providing a secure digital environment designed to support the unique and developing needs of Data Monitoring Committees (DMCs),” said Tai Xie, Ph.D., CEO and Founder of CIMS Global. “By integrating real-time statistical visualization, study-specific oversight, and GenAI-enhanced review capabilities, we aim to enhance the safety, efficiency, and scientific integrity of clinical trial oversight. DMC-HUB is an extension of our drive and commitment toward clinical excellence.”
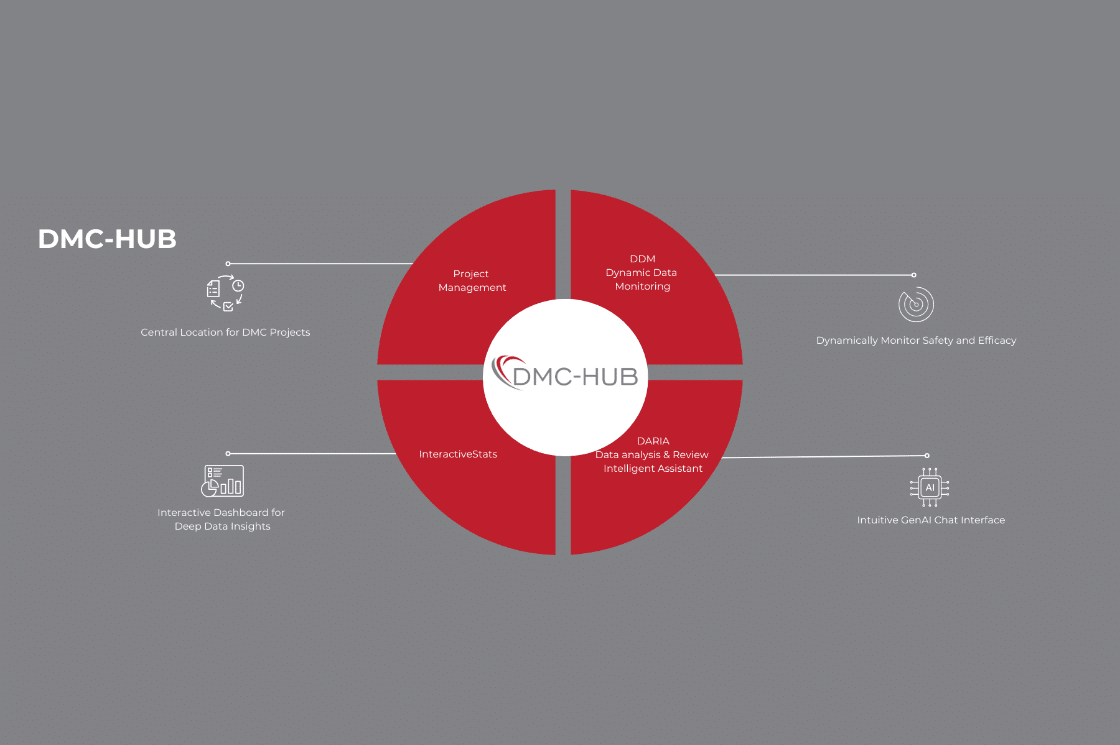
At the heart of DMC-HUB is a suite of interconnected tools:
DMC-Project Management – Study-Specific Oversight
Facilitates organized DMC operations with:
- Predefined Workflows: Structured paths for consistent decision-making
- Secure Document Sharing: Role-based folders and data protection
- Schedule and Communication Tools: Access to meeting logistics and reports in one convenient location
- Secure, Role-Based Access: Role-specific folder access ensures that only authorized personnel can access sensitive data, significantly reducing potential security risks.
DDM (Dynamic Data Monitoring) – Real-Time Statistical Visualization
The DDM system applies innovative statistical techniques to visualize cumulative trial data dynamically through a unique clinical trial “Radar Screen,” categorizing data trends as Favorable, Hopeful, Unfavorable, or Futility.
Key Features:
- Automatic calculation of Z-values, B-values, and conditional power
- Sample size re-estimation and futility assessment
- Visualization of trial progression and outcome trends
- Reconstruction of completed trials for retrospective diagnosis
InteractiveStats – Integrated R Shiny Applications
DMC-HUB incorporates Interactive Stats, a suite of R Shiny applications seamlessly integrated into the platform to support data-driven decision-making and enhance stakeholder communication.
Key Capabilities:
- Creating statistical analysis output into an interactive dashboard
- Generating executive summaries for DMCs
- Producing open-session slides for sponsors
DARIA (Data Analysis & Review Intelligent Assistant) – GenAI-Enhanced Review
Now integrated within DMC-HUB, DARIA is a beta-stage, GenAI-powered chatbot designed to assist DMC statisticians and reviewers by enabling intelligent, interactive exploration of clinical datasets.
Key Capabilities:
- Upload and review data from multiple formats, including: .sas7bdat, .pdf, .csv, .xml
- Narrative generation for summary documents and publication-ready outputs
- Chat continuity to support contextual, persistent analysis during review
- Built-in data privacy safeguards and session-level security
- Real-time response optimization using advanced LLM and intelligent decision-making
Security & Risk Management
CIMS-Global prioritizes data integrity and regulatory requirements:
- No data is stored or shared outside of the DMC-HUB environment
- DARIA sessions are fully secure and deleted upon logout
- Risk management plans include an assessment of AI model failure, bias, drift, and interpretability prior to full release
Join the Future of DMC Oversight!
Contact us to learn more
📩 contact@cims-global.com