
R Services
We provide R-based services for Clinical Trials
As a leading R technology company in the clinical trials space, CIMS Global provides advanced statistical programming solutions tailored for the pharmaceutical and biotech industries. Whether you need contractor-based services or project-based support, we tailor our solutions to your needs.
Services
Experienced R Programmers
(Contractor Provider Model)
Skilled professionals available to meet your
project needs.


R Environments in Compliance with Regulatory Requirements
Building or validating secure and compliant R environments.
R Shiny Applications and R Package Development
Custom solutions to enhance data visualization
and usability.


SDTM/ADaM Creation Using R
Efficient data standardization for clinical trial
submissions.
Statistical Analysis of Safety &
Efficacy for Clinical Trials
Supporting study reporting (TFLs) and critical
decision-making.


R-Based Data and Programming
Packages for Regulatory Submission
Ensuring a smooth and compliant submission
process.
SaaS Services Using Our Innovative R Tool
Featuring InteractiveStats and Stats2CSR for
streamlined analysis and reporting.

InteractiveStats
Transform your data review process with Interactive Stats, an advanced dashboard designed for seamless data review of ongoing clinical trials.
- Dynamic Monitoring: Gain real-time insights with an interactive interface tailored for efficient statistical analysis.
- Powered by R Shiny: Leverage robust, open-source technology for instant data visualization and review.
- Enhanced Decision Support: Deliver deeper insights to support Medical Monitors or Data Monitoring Committee’s (DMC) recommendations with confidence.
Experience streamlined trial oversight with InteractiveStats – where precision meets efficiency.
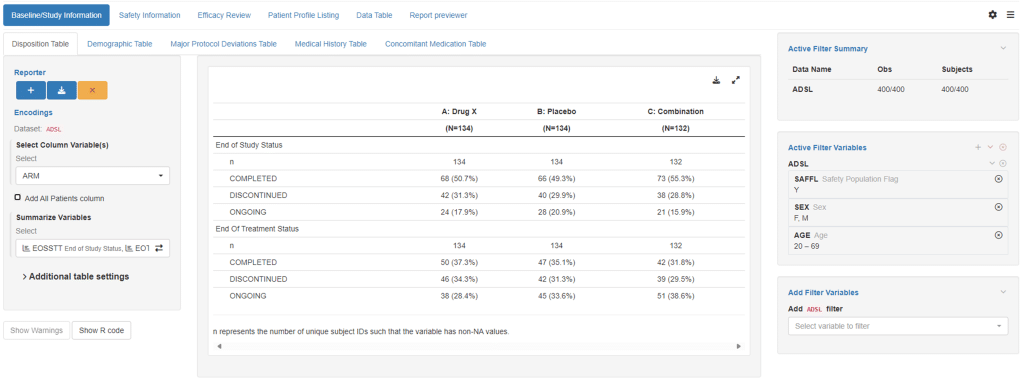
Stats2CSR
Streamline your medical writing process with our advanced workstation designed specifically for medical writers. This powerful tool enables seamless review, editing, and generation of reports while directly interacting with results from TFLs (Tables, Figures, and Listings). Incorporate insights effortlessly into customizable templates that can be updated and reused for consistent, repeatable workflows.

Let Us Help With Your R Programming
With our deep expertise in biostatistics and R technology, we help accelerate drug development while delivering robust, reliable results.

