This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Author: Carolina Simon
-
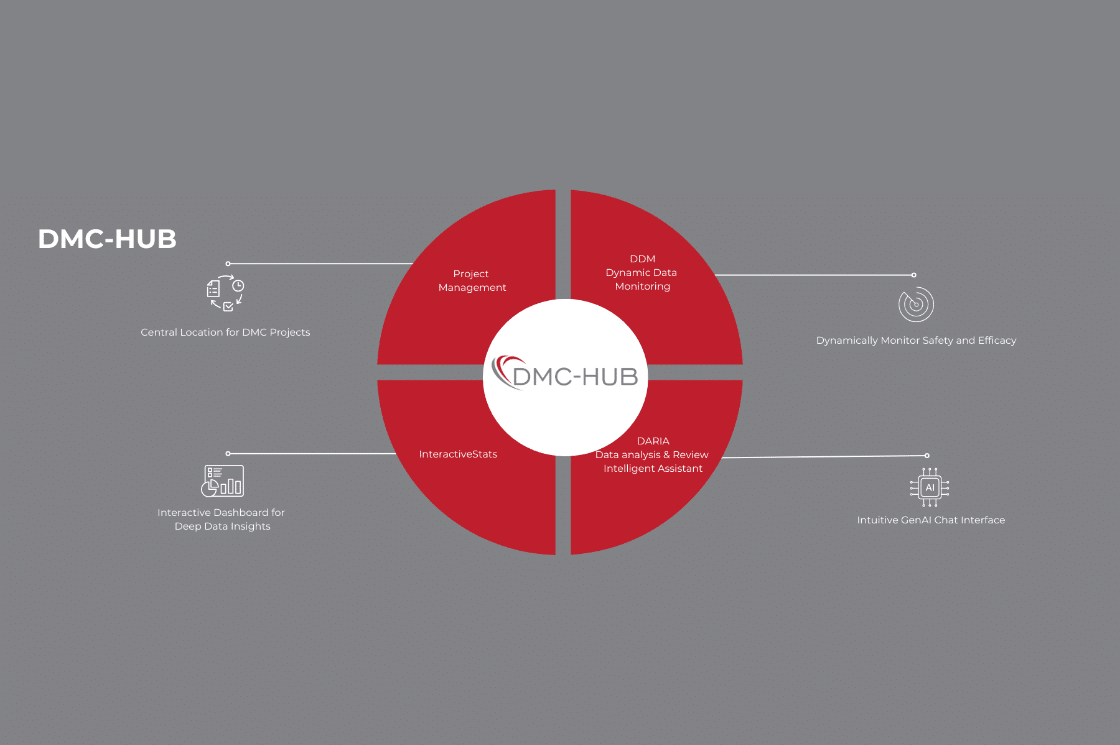
CIMS Global Launches DMC-HUB 1.0: A Centralized Ecosystem for Modern Data Monitoring Committees
CIMS Global has launched DMC-HUB 1.0, a centralized digital ecosystem built to meet the complex and evolving demands of Data Monitoring Committees (DMCs) in today’s clinical trials. Featuring integrated project management, real-time statistical visualization, interactive dashboards, and GenAI-powered review tools, DMC-HUB sets a new standard for secure, efficient, and data-driven DMC operations.
-

Dr. Tai Xie is Invited to Speak at the Upcoming Digital Technology in Clinical Trials Conference
Dynamic Data Monitoring for Ongoing Clinical Trials Digital Technology in Clinical Trials Conference October 13-14, 2022 Hyatt Regency Orange County, California
-

Dr. Peng Zhang is Presenting at the Joint Statistical Meeting in Washington, D.C.
Monitoring Ongoing Clinical Trials Under Fractional Brownian Motion with Drift Peng Zhang* and Tai Xie and Yong Lin and Weichung Joe Shih and K. K. Gordon Lan Abstract: Over the past decades, there has been lots of methods developed for monitoring clinical trials and adaptive design under Brownian motion structure. Those approached were built under…
-

Invited talks at ASA NJ Chapter for the 2020 Spring Symposium
Monitor On-Going Clinical Trials with a Dynamic Procedure Dr. Joe Shih, Professor at Rutgers, Sr. Advisor of CIMS Tai Xie, Ph.D., CEO of CIMS Global Webinar Date & Time Speaker Title Length Bio/Abstract 6/26/2020 10:30AM -12:00PM Dr. Tai Xie Monitor On-Going Clinical Trials with a Dynamic Procedure 45 mins Received Dr. Joe Shih DDM…
-

DDM has Been Used by the Remdesivir Trial in China
DIA Therapeutic Innovation & Regulatory Science Publication: Data Monitoring for the Chinese Clinical Trials of Remdesivir in Treating Patients with COVID‑19 During the Pandemic Crisis Weichung J. Shih · Chen Yao · Tai Xie Two phase-III, double-blind, randomized clinical trials of remdesivir plus SOC (standard of care) versus placebo plus SOC have been conducted in…
-

Dr. Tai Xie Discusses DDM at JSM
CIMS Global CEO Dr. Tai Xie presented the concept of DDM – Dynamic Data Monitoring – at the JSM conference in Denver, Colorado on July 27, 2019. DDM is an innovative design that combines Dynamic Adaptive Design (DAD) with a data-guided analysis for timing the SSR. The DDM process covers: Continuously monitoring on-going data Data-guided…
-

Dr. Tai Xie Presents at the Innovative Trial Design Symposium
Recently, CIMS Global founder Dr. Tai Xie presented at the Innovative Trial Design Symposium in Beijing. His talk, titled Dynamic Adaptive Design and Dynamic Data Monitoring, focused on how machine learning and artificial intelligence can reshape the future of clinical trials. By using a new approach called Dynamic Adaptive Design, (DAD), supported by real-time patient…
-

Introducing the Innovative Trial Design Group (ITDG)
About the Innovative Trial Design Group (ITDG) CIMS, the pioneer of dynamic trial design (DTD) and dynamic data monitoring (DDM), announced new services offered to the pharmaceutical and biotech industry. The new services include, but are not limited to, the following: Innovative Trial Design Group Sequential Design Adaptive Sequential Design Dynamic Adaptive Design Phase II/III…
-

CIMS Founder Dr. Tai Xie to Speak at DIA China
CIMS Global founder Dr. Tai Xie, an industry pioneer focused on reshaping the future of clinical trials through the convergence of EDC and IWRS, statistical modeling and machine learning, will be speaking at DIA China on May 22, 2019. Dr. Xie’s talk, titled Use of EHR Data in Clinical Trials (Understanding FDA’s Recent Guidance), will…
-

CIMS Adds Clinical Trial Design Expert to the Team
CIMS Global, the pioneer of dynamic trial design (DTD) and dynamic data monitoring (DDM), has added another clinical trial expert to its advisory board. Kuang-Kuo Gordon Lan will serve as a Sr. Advisor and received his PhD in Mathematical Statistics (1974) from Columbia University and is serving as CIMS Senior Statistical Advisor. Dr. Lan has published…

